The altered levels of PABPC1 suggested that it may facilitate a dynamic response to an increased workload being placed on the heart. Therefore, more work is needed to clarify the mechanism by which PABPC1 mRNA translation and poly(A) tail length is controlled in cardiomyocytes. This is surprising since studies in multiple cell types have shown that sequences within the 3’untranslated region normally regulate poly(A) tail length. found that the 3’untranslated region of PABPC1 mRNA is not important for its regulated translation. It was already known that the translation of PABPC1 mRNA was controlled by two mechanisms (reviewed in Eliseeva et al., 2013), but uncovering that its poly(A) tail length is also regulated further underscores how important it is to control cellular levels of PABPC1. Intriguingly, the length of the poly(A) tail of PABPC1 mRNA and its translation were increased in experimental models of both physiological hypertrophy (induced by swimming) and pathological hypertrophy (induced by narrowing of the aorta to mimic high blood pressure), leading to an increase in PABPC1 levels. also found that this reduction in translation coincided with a shortening of the 3’ poly(A) tail of PABPC1 mRNA: poly(A) tails are critical for efficient translation but their length can be exquisitely regulated by a number of mechanisms (reviewed in Charlesworth et al., 2013). show that PABPC1 is abundant in the neonatal heart, but is only present at very low levels in the adult heart, and they go on to show that the low levels of PABPC1 in the adult heart are due to reduced translation of PABPC1 mRNA. Protein synthesis is surprisingly low in the adult heart, perhaps reflecting the relatively low level of cell turnover there. However, it is best characterized as a factor that is required for efficient messenger RNA (mRNA) translation and to maintain mRNA stability, and it is generally considered to be an abundant and essential protein ( Smith et al., 2014). 
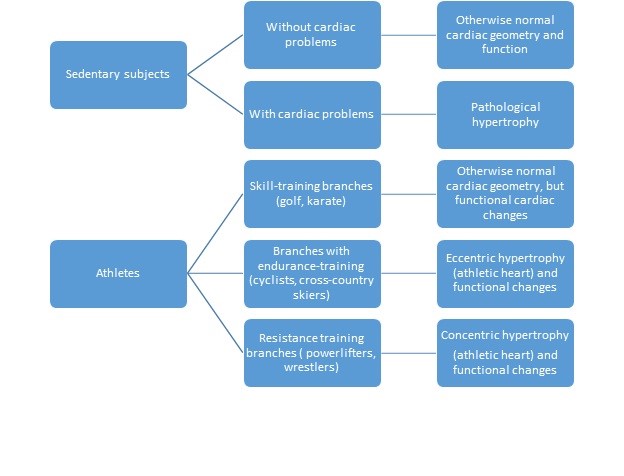
PABPC1 (commonly known as PABP1) is one of the 1000+ RNA-binding proteins found in humans, and has multiple functions in controlling the synthesis of new proteins (reviewed in Smith et al., 2014). During cardiac hypertrophy, longer poly(A) tails are associated with more efficient translation of the messenger RNA for PABPC1. This raises the question (bottom right) of whether PABPC1 function in other types of cardiac cells (such as endothelial cells or fibroblasts) may be important in the induction of pathological cardiac hypertrophy. However, a number of features (such as cardiomyocyte growth, higher protein synthesis and higher levels of PABPC1) are common to both physiological and pathological cardiac hypertrophy. induced by pregnancy or endurance exercise) but not (red broken arrow) pathological cardiac hypertrophy (e.g. The temporal overexpression of PABPC1 in adult cardiomyocytes leads to a form of cardiac hypertrophy (top right) that resembles (green arrow) physiological cardiac hypertrophy (e.g. In a normal adult heart, cardiomyocytes (CM) show little growth or turnover, and protein synthesis and PABPC1 levels are low. Now, in eLife, Auinash Kalsotra of the University of Illinois and co-workers – including Sandip Chorghade and Joseph Seimetz as joint first authors – reveal an unexpected role for an RNA-binding protein called PABPC1 in cardiac hypertrophy ( Chorghade et al., 2017). Although cellular signalling pathways and gene transcription change in distinct ways during 'physiological' and 'pathological' cardiac hypertrophy, both require de novo protein synthesis in order to increase cardiomyocyte size ( Figure 1 reviewed in Heineke and Molkentin, 2006). Cardiac hypertrophy can also be pathological in nature – for example, when it is caused by long-term hypertension. In adulthood cardiac hypertrophy can occur as a physiological adaptation to an increased need to pump blood following sustained athletic training or during pregnancy (reviewed in Maillet et al., 2013). Thereafter, cardiac growth is achieved mainly by increases in the size of cardiomyocytes in a process known as hypertrophy. The proliferation of cardiomyocytes is key to the growth of the heart before birth, and also soon after birth, but most cardiomyocytes lose their capacity to proliferate after the early post-natal period in both mice ( Soonpaa et al., 1996) and humans ( Bergmann et al., 2015). Muscle cells known as cardiomyocytes make up 25–35% of all cells in the heart, with the remainder largely being vascular endothelial cells and fibroblasts ( Pinto et al., 2016). The mammalian heart is a muscular pump that handles about five litres of blood per minute in the average human. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
